 
The molar mass is calculated in two ways. Avogadros number tells the number of particles present per mole. Our final answer is expressed to four significant figures. The periodic table provides the atomic masses used in the calculation of the molar mass. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals. 3.987 molAl × 26.98gAl 1 molAl 107.6gAl 3.987 mol Al × 26.98 g Al 1 mol Al 107.6 g Al. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The unity for atomic mass is gram per mol. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in Au: Molar Mass (g/mol) Au (Gold) 1 × 196.966569 196.966569. The lightest chemical element is Hydrogen and the heaviest is Hassium. 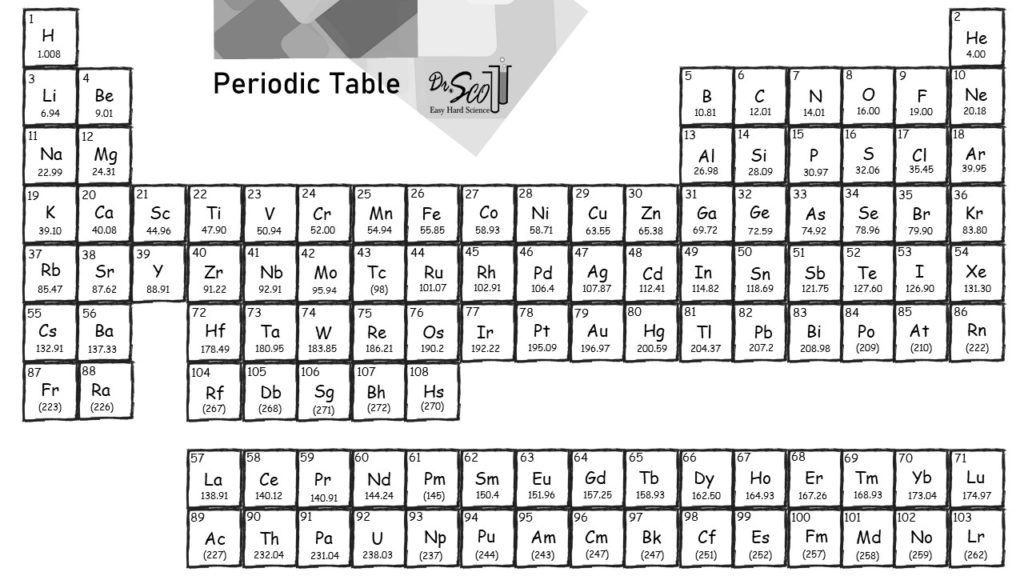
The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). This list contains the 118 elements of chemistry. Separation and Concentration Purification RequestĬhemical elements listed by atomic mass The elements of the periodic table sorted by atomic massĬlick on any element's name for further information on chemical properties, environmental data or health effects. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element.For instance, the relative atomic mass of zirconium (Zr) is 91.22. This is usually located under the symbol and name of the element. To find an element’s molar mass, start by checking the periodic table for the relative atomic mass of the element. 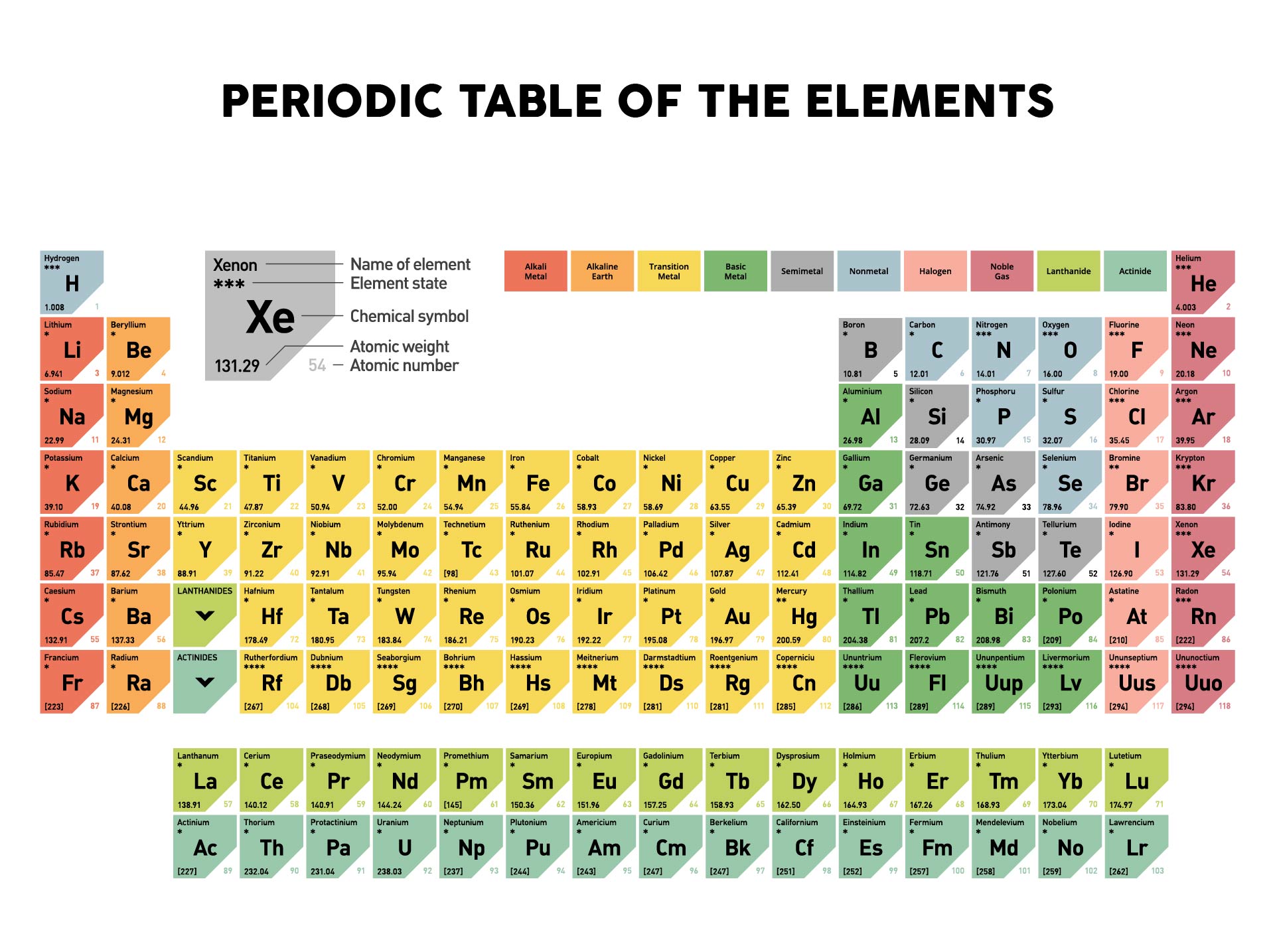
Plant Inspection & Process Optimalisation Molar mass is the mass in grams of 1 mole of any given substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
